ABSTRACT
Transesterification reactions are fundamental in various industrial processes for producing esters, yet conventional acid catalysts often lead to undesirable side reactions, product discoloration, and alcohol degradation. This study investigates the catalytic performance of Dorf Ketal Tyzor® organic titanates, highlighting their distinct advantages in promoting efficient and clean transesterification processes. Tyzor® organic titanates facilitate high-yield ester synthesis with minimal side reactions, resulting in products of superior color and odor. Notably, Tyzor® TPT emerges as the most active catalyst for carboxylic ester transesterification, demonstrating significantly faster reaction rates and higher conversions compared to sulfuric acid and p-toluenesulfonic acid, as exemplified by the synthesis of n-decyl acrylate. A major benefit of titanate catalysis is the preservation of alcohol quality, enabling the efficient recycling of recovered alcohols, thereby enhancing process sustainability. Furthermore, these catalysts prevent color buildup in the final product, crucial for demanding applications. Organic titanates are soluble and can be easily removed post-reaction through hydrolysis and filtration. Optimal reaction conditions typically involve moderate temperatures around 100 °C, with continuous removal of the byproduct alcohol to drive the reaction to completion. The versatility and efficacy of Tyzor® organic titanates make them a preferred choice for a wide range of industrial esterification and transesterification systems, offering improved product quality, increased yields, and reduced environmental impact.
DESCRIPTION
Dorf Ketal Tyzor® organic titanates are valuable catalysts in a number of chemical reactions. They are particularly useful in esterifications and transesterifications, because the catalysis is accomplished with a minimum of undesirable side reactions and results in a high yield of esters with good color and odor. For example, esters can be made from carboxylic acids and secondary alcohols with only a minor amount of olefin formation, in contrast to conventional acid catalysis, which produces substantial amounts of olefins.
Titanates are particularly useful for reactions involving unsaturated reactants and amino alcohols. Similarly, titanates are condensation catalysts in the manufacture of polyesters. Organic titanates are also useful for the catalysis and cross-linking of a wide variety of other resin types, such as polyolefins, polyurethanes, epoxies, phenolics, and silicones. Organic titanates may be used as catalysts for aldol condensations and in Meerwein-Ponndorf-Verley reductions.
TRANSESTERIFICATION
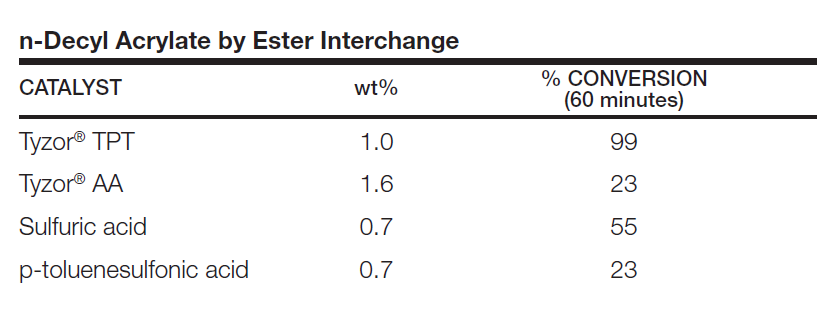
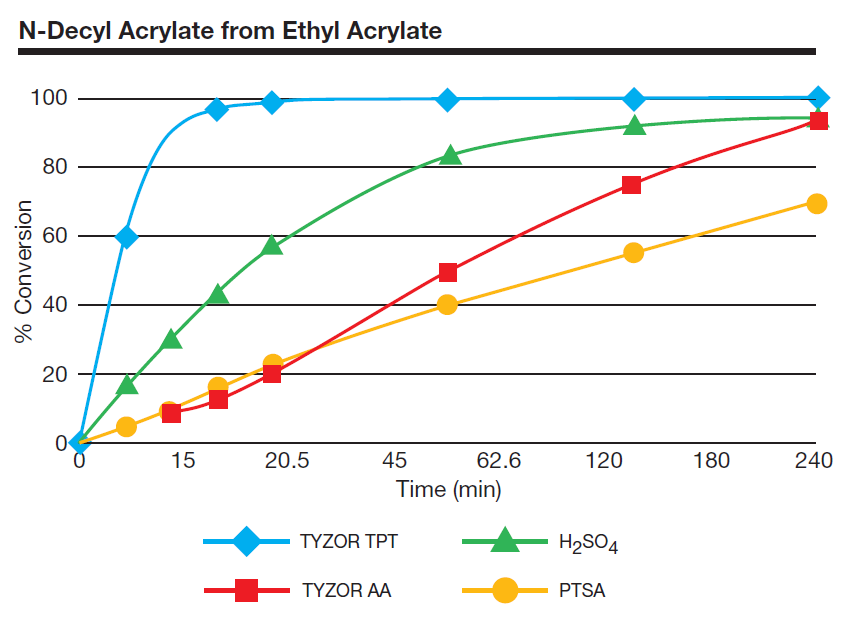
Organic titanates often give faster reaction rates and higher yieldsthan conventional acid catalysts. In the synthesis of n-decyl acrylate by the transesterification of ethyl acrylate with n-decyl alcohol, Tyzor® TPT clearly is the catalyst of choice as compared with sulfuric acid and p-toluenesulfonic acid.
A major advantage of using organic titanates is the quality of the excess alcohol recovered from the reactions. Whereas acidcatalyzed methods often cause extensive degradation of the alcohol during the reaction, titanate catalysis avoids this degradation, allowing recycling of recovered alcohols.
Equally important is the ability of organic titanate catalysis to avoid color buildup in the product and to provide high-purity esters suitable for the most demanding applications.
In reactions where color formation is a persistent problem even with titanates, several methods have been reported to minimize it, including the use of phosphorous compounds,1,2 mixed metal catalysts,3-5 organic amine co-catalysts,6-8 and titanium chelates.9-11
Titanates are superior catalysts in certain cases where color is a problem due to raw materials quality. Using organic titanates in the manufacture of trimellitate esters, for example, provides improved color in addition to the advantages of higher yield, decreased cycle time, and increased product purity.
CATALYST SELECTION
Tyzor® TPT contains the most active titanium per unit weight, and is the most active catalyst for transesterification of carboxylic esters. Tyzor® TPT is sensitive to water and is highly reactive with alcohols, acids, or other active hydrogen compounds, and may lose isopropyl alcohol quickly by exchange reactions. In most transesterifications, the first reaction is exchange of the isopropyl group with the most active and available materials. Chelates and higher alkyl titanates, such as Tyzor® TNBT and TOT, are more stable but are far less active for transesterification and require higher temperatures for high conversion rates.
Titanate catalysts are soluble and may therefore be added to the reaction mixture at any point in the process. It has been reported that adding an organic titanate catalyst late in a reaction may be beneficial, both in efficiency of catalyst usage and in product quality.
When an organic titanate catalyst is to be evaluated, all starting materials should be thoroughly dry before the titanate catalyst is added. Drying by azeotropically distilling the water with a suitable solvent is often convenient. About 0.5 to 1.0 wt% of organic titanate, based on the weight of the total product expected, is suggested as a trial catalyst level. In commercial practice, about 0.2 wt% of titanate catalyst is used, based on the weight of ester.
In transesterification reactions, temperature requirements are usually moderate, around 100 °C (212 °F). In transesterification it is particularly important that the alcohol resulting from the reaction be removed as rapidly as it is formed to drive the reaction to completion. This is especially true when methanol is produced. A polymerization inhibitor and nitrogen blanket may be desirable if a sensitive ester is being prepared (e.g., an unsaturated ester).
There are many examples of the uses of organic titanates in industrial esterifications and transesterifications.12-21 Some common reaction systems include: phthalic anhydride and 2-ethylhexanol, isophthalic or terephthalic acid and ethylene glycol, and dimethyl terephthalate and ethylene glycol.
Below is an example of a transesterification using various catalysts. There is no special relationship between the amounts of the various catalysts used.
REMOVAL OF CATALYST
Titanate catalysts are generally soluble in product esters but are easily removed by conversion to insoluble hydrolysis products, which are then filtered. Titanates may be hydrolyzed using a variety of agents, including aqueous caustic or lime when very low acidity or assistance in color treatment is desired. Generally, agitation with a small amount of water for a period of time before steam stripping is sufficient for efficient hydrolysis of the catalyst. The resulting titanium oxides are crystalline solids, highly insoluble, and easily removed by filtration.
REFERENCES
1. Union Carbide, US 4,424,140-1/3/84
2. Dynamit Nobel, GER 3,300,315-7/12/84
3. ICI, US 3,965,071-6/22/76
4. Rohm and Haas, US 4,356,299-10/28/82
5. Teijin Ltd., J 71-28,788-8/20/71
6. Teijin Ltd., J 136,294-11/14/77
7. Teijin Ltd., J 78-85,891-7/28/78
8. Teijin Ltd., J 78-39,392-4/11/78
9. Teijin Ltd., J 77-148,593-12/9/77
10. Teijin Ltd., J 76-41,792-3-4/8/76
11. Teijin Ltd., J 86-20,567-8-5/22/86
12. B.F. Goodrich, US 3,056,818-10/2/62
13. U.S. Steel, US 3,106,570-10/8/63
14. M.P. Stevens and J.D. Gardner, Indus. Eng. Chem. Process Design and Development, 4, No. 1, pp 67-71 (1/65)
15. Martin Marietta, GB 991,020-5/5/65
16. Leslie C. Case, US 3,382,217-5/7/68
17. Owens Corning Fiberglas, US 3,674,724-7/4/72
18. Dynamit Noble A.-G. GER 1,933,687-1/14/71
19. DuPont, US 2,822,348-2/4/58
20. Koppers, US 3,321,552-5/23/67
21. VEB Deutsches Hydroiewerk Rodleben, GER 2,009,209-5/19/71



